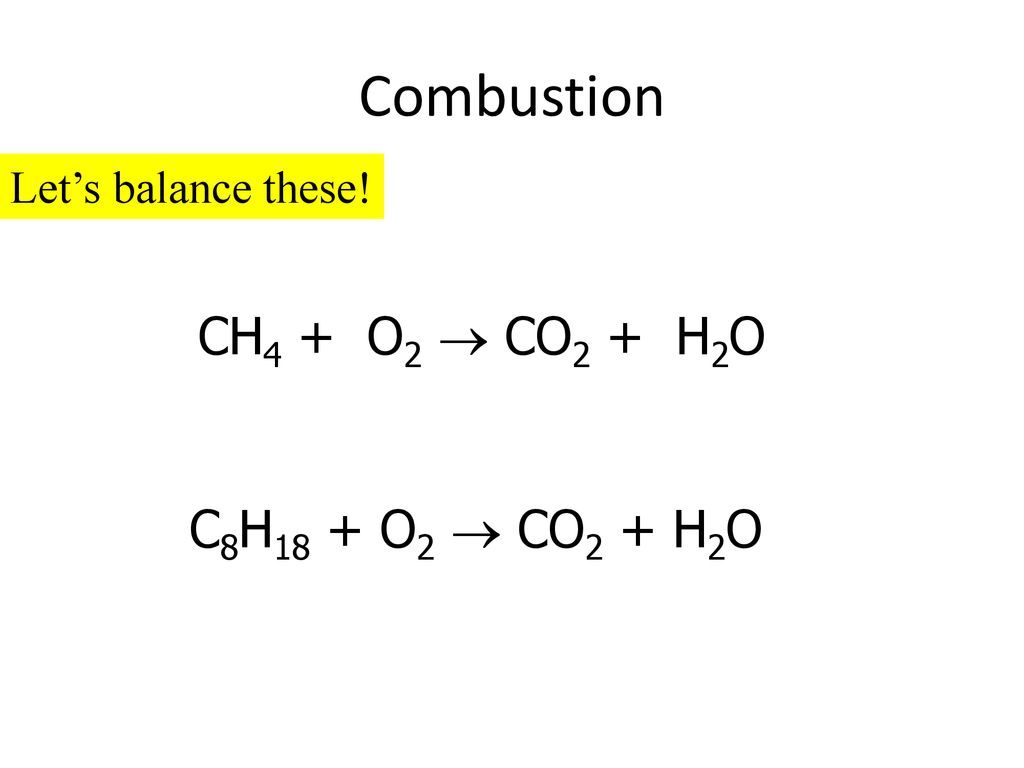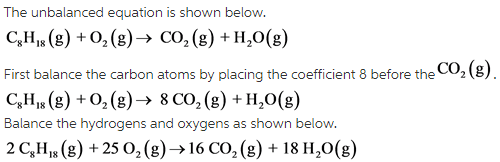
Determine the balanced chemical equation for this reaction. C8H18(g)+O2 (g)→CO2(g)+H2O(g) - Home Work Help - Learn CBSE Forum

Determine the balanced chemical equation for this reaction. C8H18(g)+O2 (g)→CO2(g)+H2O(g) - Home Work Help - Learn CBSE Forum
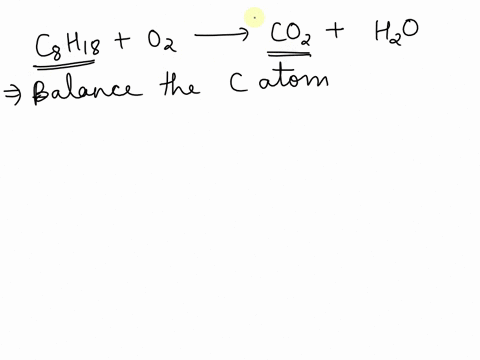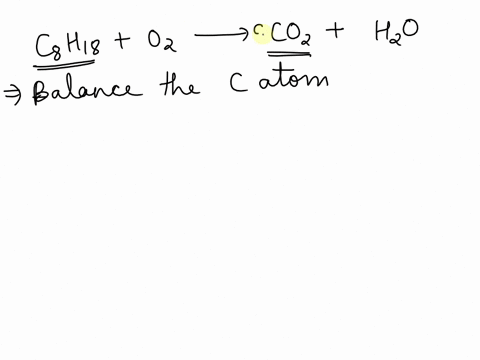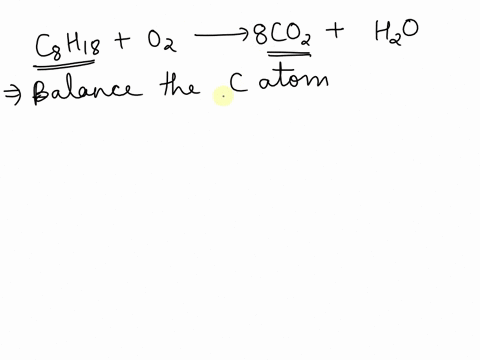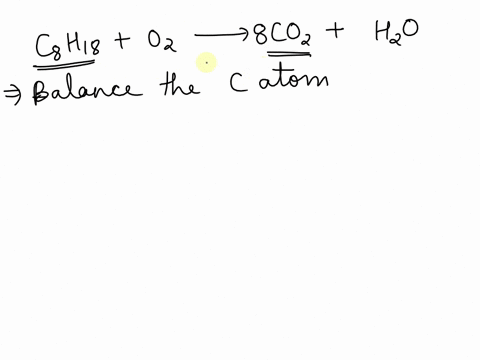
SOLVED: Balance the following equation C8H18 + O2 –>CO2 + H2O and tell me how why you did what you did, please
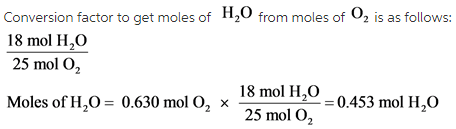
Determine the balanced chemical equation for this reaction. C8H18(g)+O2 (g)→CO2(g)+H2O(g) - Home Work Help - Learn CBSE Forum
![SOLVED: Calculate AHPrxn for the combustion reaction of octane (C8H18) shown below C8H18(l) + 25/2 O2(g) -> 8CO2(g) + 9H2O(l), given the following in kJ/mol: ΔHf[C8H18(l)] = -249.3 kJ/mol, ΔHf[CO2(g)] = -393.9 SOLVED: Calculate AHPrxn for the combustion reaction of octane (C8H18) shown below C8H18(l) + 25/2 O2(g) -> 8CO2(g) + 9H2O(l), given the following in kJ/mol: ΔHf[C8H18(l)] = -249.3 kJ/mol, ΔHf[CO2(g)] = -393.9](https://cdn.numerade.com/ask_images/5ce4062f3cf748cda28880a907be3aa1.jpg)
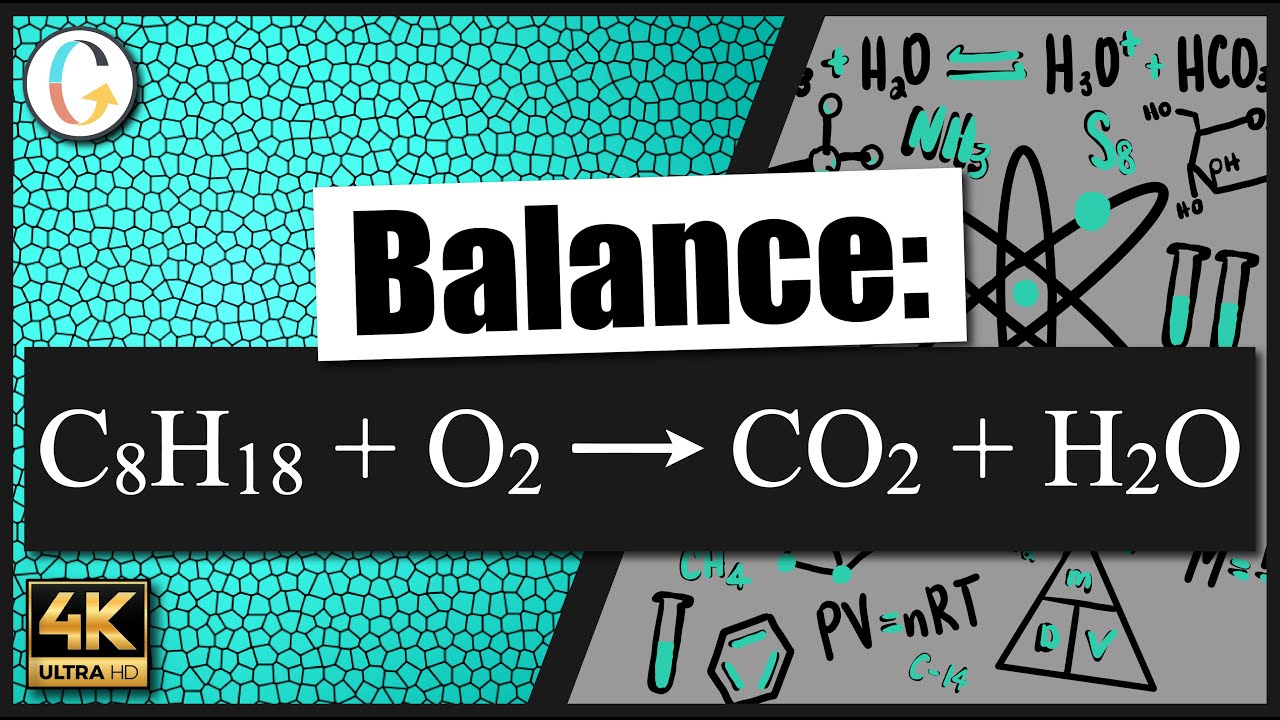
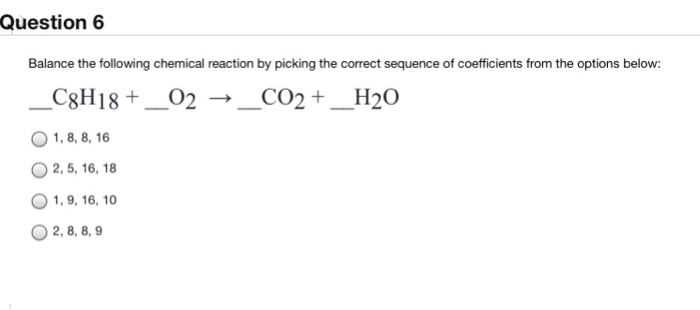
















_1.jpg)